
- Enter the desired initial concentration.
- Enter the stock solution concentration and select the appropriate unit.
- Enter the name of the diluent (solution used to dilute the stock solution).
- Serial dilutions can be calculated either using a starting concentration and dilution factor OR a concentration range.
- Select the method for calculating the serial dilution.
- The tool below can be used to create a protocol for preparing a serial dilution from a stock solution. For instance, creating a two-fold dilution with a starting concentration of 10 µM yields the following concentrations: 10 µM, 5 µM, 2.5 µM, 1.25 µM, etc. Thus, when creating standards for a given assay, it is often necessary to prepare the standards using a serial dilution.Ī serial dilution is a sequence of dilutions created using the same dilution factor. For many quantification assays, the linearity range is logarithmic in nature. This represents the range of quantities over which response values are directly proportional to changes in the target of interest. Standards for the calibration curve are typically chosen such that they span the linearity of a given assay. From this curve, the quantity of target in a sample can be calculated.

These standards are used to create what is known as a calibration curve, or standard curve.
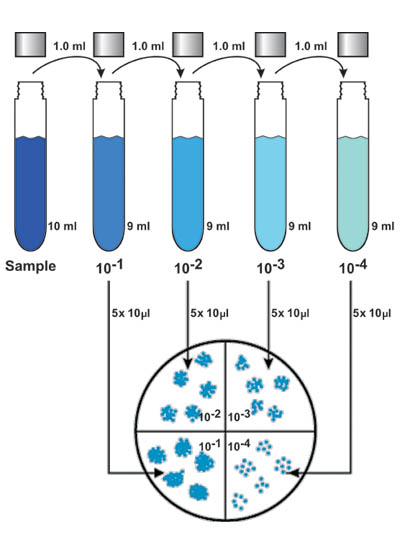
For many quantification assays, a set of standards must be run alongside test samples in order to calibrate an experiment properly.

 RSS Feed
RSS Feed
